Citric Acid | Vibepedia
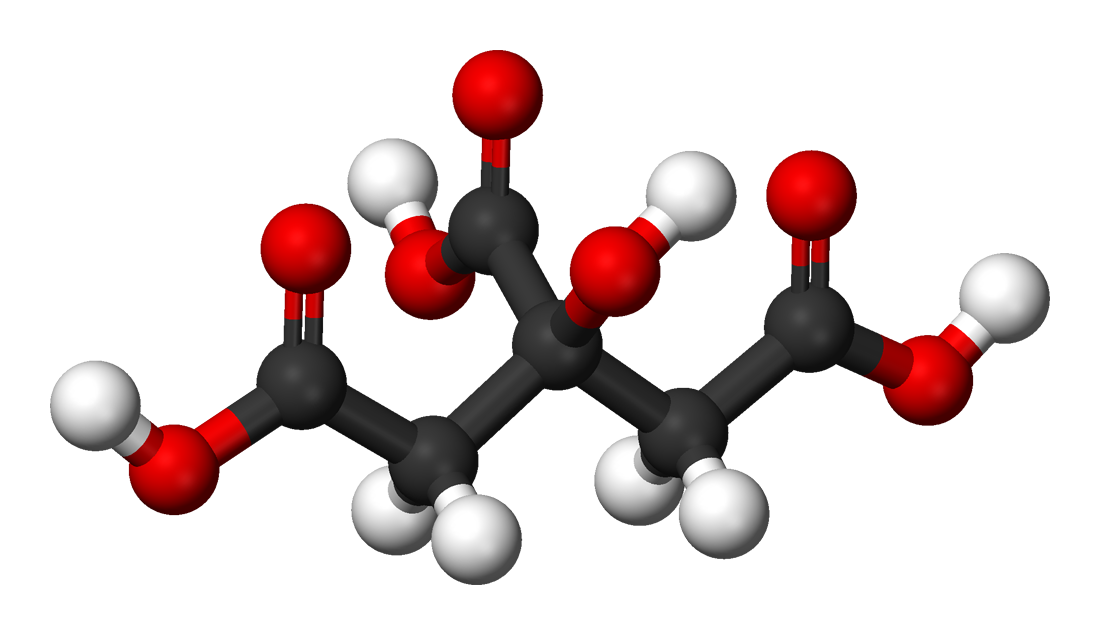
Citric acid is naturally abundant in citrus fruits. It is a critical intermediate in the citric acid cycle, the metabolic pathway essential for energy…
Contents
Overview
The story of citric acid begins not with industrial synthesis, but with the tang of citrus. While its presence in lemons and limes was known for centuries, its isolation and characterization are credited to Swedish chemist Carl Wilhelm Scheele, who first crystallized it from lemon juice in 1784. For over a century, this crystallization from fruit juice remained the primary method of production, a laborious and expensive process. The true industrial revolution for citric acid began in the early 20th century with the discovery that certain microorganisms, notably strains of Aspergillus niger, could efficiently produce citric acid through fermentation. This breakthrough, pioneered by researchers like James Currie in the 1910s, shifted production from agricultural extraction to biotechnological manufacturing, paving the way for its widespread commercial availability and diverse applications.
⚙️ How It Works
Citric acid functions through its molecular structure: a three-carbon chain with a hydroxyl group and two carboxyl groups. This trifunctional nature allows it to act as a weak acid, donating protons (H+) in aqueous solutions, which is responsible for its characteristic sour taste and its ability to lower pH. As a chelating agent, its carboxyl groups can bind to metal ions, effectively sequestering them. This property is crucial in preventing metal-catalyzed oxidation, which can degrade food products and pharmaceuticals. In biological systems, it's a central player in the citric acid cycle (also known as the Krebs cycle), where it facilitates the oxidation of acetyl-CoA, releasing energy in the form of ATP and reducing equivalents for cellular respiration. Its ability to participate in these biochemical reactions and chemical interactions makes it incredibly versatile.
📊 Key Facts & Numbers
The global citric acid market is colossal, with annual production exceeding 2 million metric tons. The vast majority of this, estimated at over 90%, is produced via Aspergillus niger fermentation, a testament to the efficiency of this biotechnological process. The market value is substantial, projected to reach over $4.5 billion USD by 2027, driven by demand from the food and beverage sector, which accounts for approximately 70% of consumption. North America and Europe are major consumers, but the Asia-Pacific region, particularly China, is the largest producer, accounting for over half of global output. The price of citric acid can fluctuate based on raw material costs, primarily corn-derived glucose or molasses, and energy prices, typically ranging from $1,000 to $1,500 per metric ton.
👥 Key People & Organizations
While Carl Wilhelm Scheele first isolated citric acid in 1784, the industrial giants of its production are modern biochemical companies. Major global manufacturers include ADM (Archer Daniels Midland), Cargill, Tate & Lyle, and numerous Chinese firms like Jilin Zhongxin Chemical Group and Shandong Heitai Biotechnology. In the realm of biochemistry, Hans Krebs is inextricably linked for elucidating the citric acid cycle in 1937, earning him the Nobel Prize in Physiology or Medicine in 1953. James Currie's early 20th-century work on Aspergillus niger fermentation was also foundational for industrial production.
🌍 Cultural Impact & Influence
Citric acid's influence permeates daily life, most notably in the food and beverage industry, where it provides the characteristic tartness in candies, sodas, and jams, and acts as a preservative to extend shelf life. Beyond taste, it's a key ingredient in bath bombs, creating the fizzing reaction with baking soda. In pharmaceuticals, it's used to mask unpleasant tastes and as an anticoagulant in blood storage. Its chelating properties make it a common ingredient in cleaning agents and detergents, helping to soften water and boost cleaning efficacy. The ubiquity of its sour flavor in everything from lemonade to sour gummies has also cemented its place in the global palate, influencing flavor profiles across countless cuisines and manufactured goods.
⚡ Current State & Latest Developments
The production of citric acid remains dominated by Aspergillus niger fermentation, a mature technology. However, ongoing research focuses on optimizing yields and reducing costs. Innovations include exploring alternative, more sustainable feedstocks beyond corn-derived glucose, such as lignocellulosic biomass or even industrial waste streams. Genetic engineering of Aspergillus niger strains aims to enhance citric acid production efficiency and reduce byproduct formation. Furthermore, there's a growing interest in developing direct enzymatic or cell-free synthesis methods, though these are still largely in the experimental phase and face significant economic hurdles compared to established fermentation processes. The market continues to grow steadily, with projections indicating a compound annual growth rate (CAGR) of around 4-5% over the next five years.
🤔 Controversies & Debates
While generally recognized as safe (GRAS) by regulatory bodies like the U.S. Food and Drug Administration, citric acid is not without its controversies. Some individuals report sensitivity or adverse reactions, such as canker sores or digestive upset, particularly with high concentrations or frequent consumption, though scientific consensus attributes these largely to the acidity rather than the molecule itself. Concerns have also been raised regarding the sourcing of raw materials for fermentation, with debates around genetically modified organisms (GMOs) used in corn production in some regions. Additionally, the environmental impact of large-scale fermentation, including water usage and waste disposal, is a subject of ongoing scrutiny and efforts towards more sustainable practices.
🔮 Future Outlook & Predictions
The future of citric acid production is likely to see a continued push towards sustainability and cost-efficiency. Research into synthetic biology and metabolic engineering holds promise for developing novel microbial strains or even entirely new production pathways that bypass traditional fermentation, potentially using less resource-intensive feedstocks. The development of circular economy models, where waste products from other industries are utilized as fermentation substrates, could significantly reduce the environmental footprint. Furthermore, as consumer demand for natural and clean-label ingredients grows, the inherent natural origin of citric acid (when produced via fermentation from plant-based sugars) positions it favorably against artificial acidulants, suggesting sustained demand and potential for new applications in areas like biodegradable polymers and advanced material science.
💡 Practical Applications
Citric acid's applications are remarkably diverse. In the food and beverage industry, it's a ubiquitous acidulant and flavor enhancer in everything from soft drinks and candies to jams and cheeses, also serving as a preservative. In pharmaceuticals, it's used in effervescent tablets, as a pH adjuster, and as an anticoagulant in blood collection tubes. The cleaning industry relies on it as a chelating agent in detergents and descalers, effectively removing mineral deposits and boosting cleaning power. It's also found in cosmetics for pH adjustment, in the textile industry for dyeing, and in metal treatment for passivation and cleaning. Even in construction, it can be used as a retardant in cement mixtures.
Key Facts
- Category
- science
- Type
- topic